Background:
Acute myeloid leukemia (AML) patients with high-risk pre-HSCT measurable residual disease (MRD, KMT2A-r, etc.) have a high relapse rate (>50%) even after allogeneic hematopoietic stem cell transplantation(allo-HSCT). In 2018, we developed two multi-institutional, single-arm, prospective phase II trials, to investigate the efficacy and safety of a novel intensified conditioning with “Mega-Dose 400mg/m 2” decitabine following allo-HSCT for high-risk MRD AML (NCT03793517).
Method:
Inclusion: 1) Detectable pre-HSCT MRD detected by recurrent genetic abnormalities (KMT2A-r, DEK, etc.) in ELN adverse-risk AML; or high-level pre-HSCT MRD in all risk groups, defined as the reduction of recurrent genetic abnormalities ≤1 log compared to initial diagnosis (RUNX1:RUNX1T1, etc.); 2) Age≥18; no HSCT history, available donor; ECOG 0-2, HCT-CI 0-3. Uniform conditioning regimen: intravenous decitabine (200mg/m 2/day) on days-12, -11, intravenous cytarabine+ busulfan+ cyclophosphamide (Bu/Cy) on days -10 to -2 (Blood. 2015;125(25):3956-62). The primary endpoint was cumulative incidence of relapse (CIR). Competing risk analysis and time-dependent Cox model were constructed in R software 4.21. Contemporary MRD+ AML patients with the same recurrent genetic abnormalities following Bu/Cy regimen or relapsed/refractory AML patients following 400mg/m 2 decitabine regimen (NCT03799224) were enrolled as nest-control to compare the post-HSCT kinetics of recurrent genetic abnormalities.
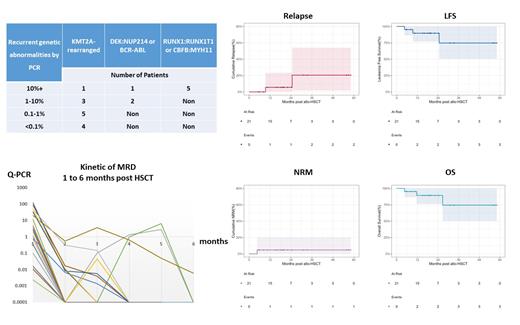
Results: Between Dec 2018-Feb 2023, twenty-one patients received allo-HSCT from HLA-matched siblings (n=2), and haploidentical donors (n=19), including 13 patients with KMT2A-r genes. Median follow-up was 19 (4.6-58) months after HSCT. The cumulative 30-day neutrophil (median 13 days) was 100.0% and 100-day platelet engraftment and 95.2% (median 21 days); all patients reached negative MRD by flow cytometry after HSCT. This conditioning regimen was well tolerated without irreversible grade III-IV toxicity peri-engraftment.
Primary endpoint: 2-year CIR was 5.6% (95%CI: 0.3-23.3). Secondary endpoint: 2-year NRM was 4.8% (0.3-20.2); 2-year LFS was 89.6% (77-100); 2-year OS was 89.3% (76.2-100). The median fold reductions (log10) of leukemia burden with recurrent genetic abnormalities were 3.6 (range 1.3-5.1) one month after HSCT (Figure).
Conclusion: Compared to Bu/Cy regimen, “Mega-Dose” decitabine (400mg/m2) is a novel conditioning regimen with improved efficacy to eradicate pre-HSCT MRD and reduce post-HSCT relapse.
Disclosures
No relevant conflicts of interest to declare.